Applications
Contactless media change & liquid evacuation with near zero, reproducible residual volume Magnetic bead* washes with magnetic carrier for 96, 384, 1536 well MTPs
Contactless media change & liquid evacuation with near zero, reproducible residual volume Magnetic bead* washes with magnetic carrier for 6, 12, 24, 48, 96 and 384 well MTPs
Compatibility
Any SBS format plate up to 17.4mm height (including 1536 well) strip or slides, custom formats programmable; dispensing into 96 and 384 well plates; 1536 well MTP option
Any SBS format plate up to 27mm height strip or slides, custom formats programmable; dispensing into 6, 12, 24, 48, 96 and 384 well plates
Centrifuge system
Liquid evacuation by centrifugation; plates positioned in symmetric rotor with well openings facing outwards to expel liquids. Rotor driven by computer controlled, high power servo motor
Residual volume
0.01μl to 5μl per well, depending on application-specific evacuation method selected
Shaking & incubation
Programmable speeds, rocking angles and time, ideal to break up liquid layers
Centrifuge specs
RPM: 3 to 3,500 RPM (= 630 g), Acceleration: <1,500 RPM/s, Deceleration:< 1,000 RPM/s
Maximum weight difference between active plate and balance plate in dry condition:
15 g @ 3,500 rpm, 20 g @ 2,000 rpm, 35 g @ 1,500 rpm, 70 g @ 1,000 rpm
Dispense system
Precision membrane pump and electromechanical valves controlled by on-board Linux CPU
Liquid inputs
Choice of 4 or 7 liquid inputs with Luer lock fittings
Specifications
(all volumes per well)
Manifold: 8, 16, or 32 needles (for 96/384/1536 well MTP)
Dispense jet position: Slanted dispenser mount to aim at well walls, plate off-set adjustable
Dispense pressure: adjustable in 5 steps, from approx. 25% to full pressure
Dispense speeds: 96w MTP: 7 sec @ 100μl; 384w MTP: 13 sec @ 50μl; 1536w MTP: 13 sec @ 8μl
Range & accuracy: 96 and 384 MTP: >1μl, < 3%CV @ 100μl; 1536 MTP: >3μl, < 5%CV @ 10μl
Manifold: 2, 3,4, 6, 8, 16 needles (for 6/12/24/48/ 96/384well MTP)
Dispense jet position: Slanted dispenser mount to aim at well walls, plate off-set adjustable
Dispense pressure: adjustable in 5 steps, from approx. 25% to full pressure
Dispense speeds: 96w MTP: 7 sec @ 100μl; 384w MTP: 13 sec @ 50μl
Range & accuracy: 6, 12, 24, 48 MTP: >1μl; 96 and 384 MTP: >5μl, < 3%CV @ 100μl;
Internal dead volume
8 ml (input connector to dispenser pin tips)
Tubes
Reagent tubes: length: 1m, Ø outside: 5mm, Ø inside: 3mm; Luer locks on both ends connect Blue®Washer input to Luer lock on GL-45 cap for reagent bottle, color-coded
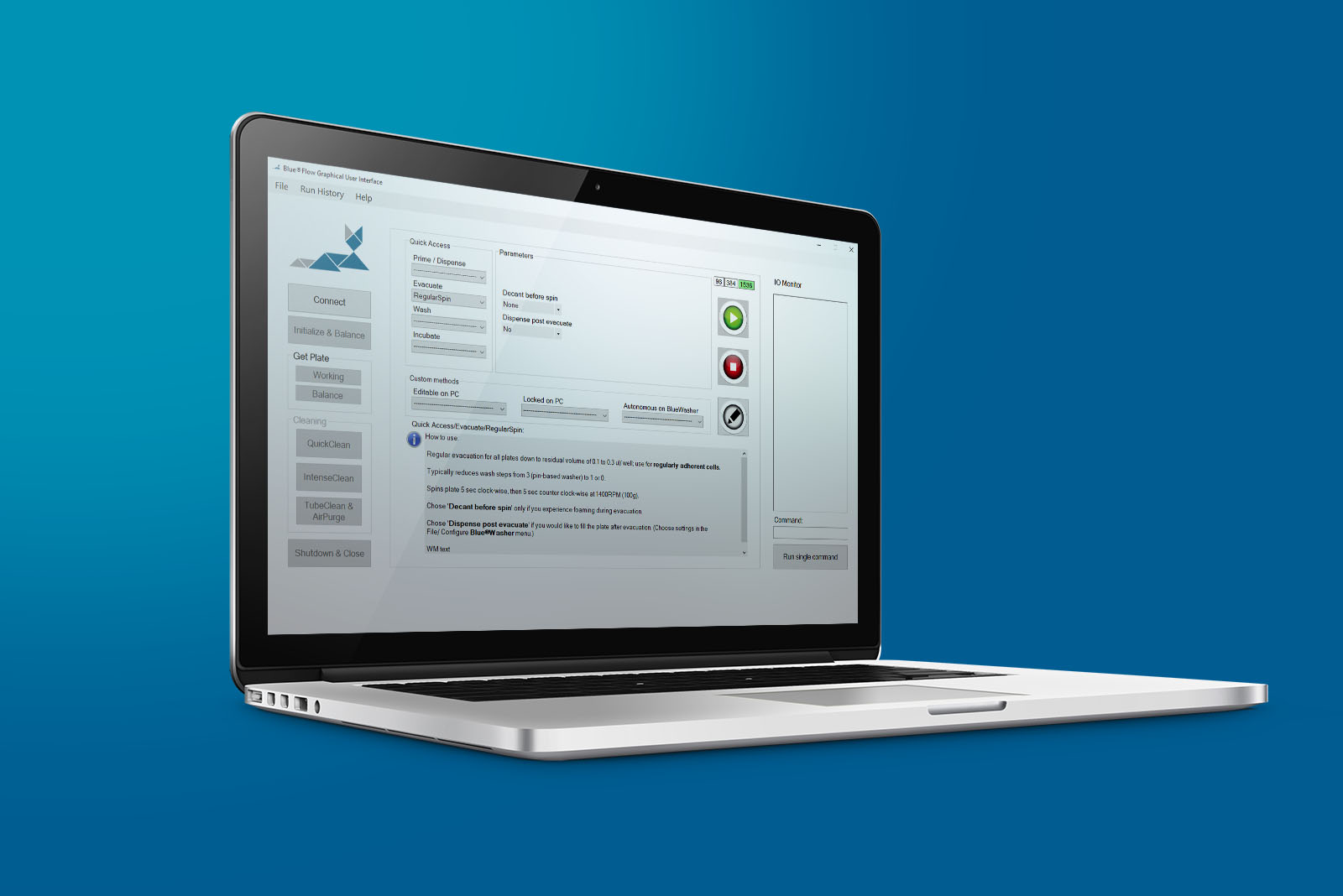
Software and operation
PC-based GUI with quick access to pre-set, application specific evacuation methods and editor to customize methods and program workflows. Autonomous mode to run methods from Blue®Washer’s internal memory
Automation
Easy plate load/ pick-up by simple robotic gripper; standard API, USB and Ethernet
Available automated plate loading via Blue®Bench; choice of plate storage in up to 5 stacks or random access hotels with auto de-lidding/ re-lidding
Safety
Centrifugation shut off in case of excessive vibration; sensor controlled interlocking door
Cleaning & decontamination
Single touch QuickClean and IntenseClean protocols with custom Blue®Daily and Blue®Intense cleaning solutions highly effective to clean and decontaminate Blue®Washer interior
Dimensions (HxWxD)
20.5 cm x 36.5cm x 69.0 cm | 8.1" x 14.4" x 27.2"
20.5 cm x 36.5cm x 69.8 cm | 8.1" x 14.4" x 27.5"
electrical and shared liquid connections on left side wall, dedicated liquid connections on right side wall
Weight
27 kg | 60 lbs
28 kg | 62 lbs
Power requirements
100 – 240 V, 50/60 Hz, 350 W, auto sensing, I max = 10A
Ambient temperature
15°C – 55°C | 60 F – 130F